The safety guidance below describes the proper laboratory practices for storing biological materials in liquid nitrogen. Hazards associated with Liquid Nitrogen include extreme cold, asphyxiation, and explosion; resulting in sample loss and personal injury. (see Cryogens and Dry Ice). The principal investigator is responsible for developing and implementing standard operating procedures (SOPs) for safe handling of biological material stored in liquid nitrogen.
Personal Protective Equipment (PPE)
Eye/face protection
An impact resistant full face shield over safety glasses or chemical splash goggles are recommended when placing or retrieving samples in liquid nitrogen. Additionally, full face protection is required when thawing cryogenic vials.

Skin Protection
A long sleeve lab coat, protective gloves that are rated for work with liquid nitrogen, pants that fully cover the legs, and closed toe shoes should be worn while handling biological samples stored in liquid nitrogen.
Equipment and Supplies
Cryogenic Vials/Gaskets
Sample storage vials should be rated for cryogenic storage; most cryogenic vials are only rated for vapor phase storage. Seals/gaskets of cryogenic vials should be sufficient to seal the vial in vapor phase (as low as -196ºC) and some materials (e.g. Thermoplastic Vulcanizates; TPV) are resilient to overtightening of lids, which promotes leaking. If liquid phase storage is required (unlikely for most biological samples) for your application, be sure to use specialized storage materials that are rated for this purpose (e.g. Cryoflex tubing).
Dewars/Vessels
Cryogenic vessels, or dewars, are rated for liquid nitrogen storage and should be used to store biological samples. The user should ensure that the dewar is undamaged to meet the original manufacturer specifications.
Practices
Cryogenic Vial Filling and Storage
Vials should only be filled to the designated fill line indicated by the manufacturer, typically no more than 85% nominal capacity, to reduce the possibility of sample expansion beyond
CAUTION:
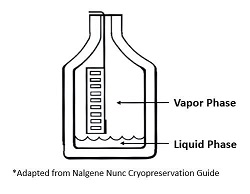
Be aware that cryogenic vials stored in the liquid phase may take on liquid nitrogen, which may result in vial rupture upon thawing leading to sample loss and injury. The risk can be reduced by moving cryovials to the vapor phase for at least 24 hours prior to removal.
Biological Sample Handling
Sample vials should be disinfected on the outside prior to being placed in liquid nitrogen. Biohazardous materials should be handled according to Biosafety in Microbiological and Biomedical Laboratories (BMBL) containment guidelines. Use tongs with insulated handles or other mechanical devices to insert and withdraw items.
Dewer/Vessel Filling
Fill Dewar/vessels to an appropriate level to maintain samples in the vapor phase while avoiding submersion of samples. When filling, insert the hose into the opening of the container and slowly open the extraction valve. Boiling and splashing always occur when charging or filling a warm container with cryogenic liquid, therefore proper PPE should be worn. When filling is complete, close the valve and remove the hose carefully as it will be extremely cold.
Decontamination
If liquid nitrogen storage vessels become contaminated due to leaking of vial contents, improper disinfection of the exterior of sample vials, or rupture of vials, then the entire storage vessel should be decontaminated with an EPA registered disinfectant after warming the vessel to room temperature.
Emergency Procedures
Before work begins, familiarize yourself with proper actions to take in the event of an exposure to liquid nitrogen or biological samples. Instructions for emergency response and injury reporting can be found at the DRS website.
References
Biosafety in Microbiological and Biomedical Laboratories (BMBL, 5th Ed.)